


In the world of ethyleneamines, selecting the right intermediate can drastically alter the performance of your final product. For specialized formulators and buyers at Bewellchem, two of the most common inquiries we receive revolve around the comparison between Ethylenediamine and Diethylenetriamine.
While they belong to the same chemical family, the What is the Difference Between EDA (Ethylenediamine) and DETA? debate comes down to molecular architecture and reactivity profiles.

At a fundamental level, the primary difference lies in their chemical structure.
● EDA (Ethylenediamine): This is the simplest polyethylene polyamine, consisting of two primary amine groups connected by an ethyl bridge. It is a diamine.
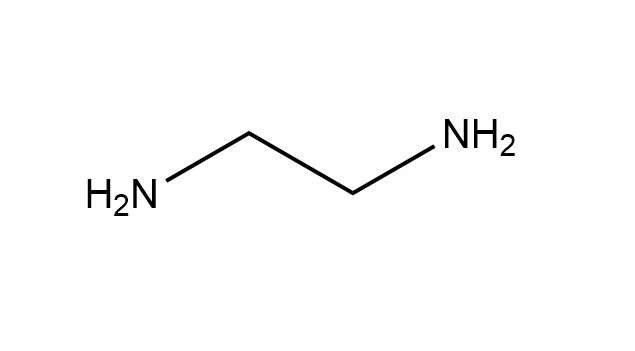
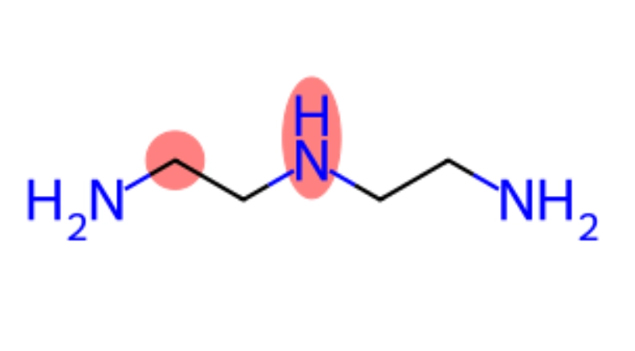
● DETA (Diethylenetriamine): This is a linear analog containing three amine groups (two primary and one secondary) linked by two ethyl bridges.
Because DETA has an additional nitrogen atom and a longer chain, it exhibits different physical properties, such as a higher boiling point and higher viscosity compared to the relatively volatile EDA.
For chemists involved in stoichiometry, the Amine equivalent weight is the most critical metric. This value represents the amount of the amine required to react with one equivalent of an epoxy group or acid.
1. EDA has a lower molecular weight (60.10 g/mol), leading to a very high amine density.
2. DETA (103.17 g/mol) provides more functional sites per molecule, which influences the cross-linking density in polymer networks.
In EDA vs DETA comparisons, EDA is often favored for its efficiency in small-molecule synthesis, whereas DETA is preferred when a slightly more flexible, yet highly reactive, backbone is required.
When it comes to curing agent selection for epoxy resins, the choice between these two polyamine differences becomes apparent in the curing speed and the brittleness of the final resin.
● EDA tends to produce very "tight" cross-linking. This results in hard, chemical-resistant coatings, but they can be prone to brittleness if not modified.
● DETA is a staple in the manufacturing of wet-strength resins for paper and as a hardener in floor coatings. Its three reactive nitrogen sites allow for a more complex three-dimensional network, offering a balance between toughness and thermal resistance.
When Ethylenediamine compared to DETA, you must consider the environmental conditions of your process. EDA’s higher vapor pressure makes it more challenging to handle in open-air applications due to its "blushing" effect and fumes. DETA, being less volatile, offers a slightly safer handling profile in industrial settings.
|
Property |
EDA |
DETA |
|
Amine Groups |
2 (Primary) |
2 (Primary), 1 (Secondary) |
|
Molecular Weight |
~60 |
~103 |
|
Boiling Point |
116°C |
206°C |
|
Key Use |
Chelating agents, bleach activators |
Epoxy hardeners, fuel additives |
At Bewellchem, we understand that navigating polyamine differences requires technical expertise and a reliable supply chain. Whether you are synthesizing EDTA, producing surfactants, or formulating high-performance epoxy systems, our team provides high-purity EDA and DETA tailored to your specifications.
Choosing the right amine is not just about the price—it is about optimizing your reaction and ensuring the longevity of your product. Contact us today to discuss your technical requirements and let us help you streamline your curing agent selection.
leave a message
Scan to wechat :

Scan to Whatsapp :
